Trends in Mortality Rates for Dilated and Hypertrophic Heart Disease in the United States From 1999 to 2023
DOI:
https://doi.org/10.65364/iomj.2026.05Abstract
Background: This study aimed to analyze U.S. nationwide mortality trends from 1999 to 2023 for the major non-ischemic cardiomyopathies—dilated (DCM) and hypertrophic (HCM) cardiomyopathy—with a focus on demographic and geographic disparities.
Methods: Mortality data for adults aged ≥25 years were extracted from the CDC WONDER database using ICD-10 codes I42.0–I42.2. Age-adjusted mortality rates (AAMRs) per 100,000 population were calculated and standardized to the 2000 U.S. population. Joinpoint regression was used to evaluate temporal trends, estimating annual percentage change (APC) and average annual percentage change (AAPC).
Results: Between 1999 and 2023, there were 101,871 DCM-related and 24,879 HCM-related deaths. The AAMR for DCM declined from 3.00 (95% CI, 2.92–3.08) in 1999 to 1.10 (95% CI, 1.05–1.14) in 2023 (AAPC –3.64%). For HCM, the AAMR decreased from 0.75 (95% CI, 0.71–0.79) to 0.35 (95% CI, 0.32–0.37) (AAPC –3.25%). Mortality rates decreased across all demographic groups but remained highest among older adults (≥85 years), men, and non-Hispanic Black individuals. Regional variation was evident: DCM mortality was lowest in the Northeast, while HCM mortality was highest in the West.
Conclusions: Mortality from both DCM and HCM declined substantially in the U.S. from 1999 to 2023.
CLINICAL RELEVANCE
This study provides the first comprehensive assessment of long-term mortality trends for dilated cardiomyopathy and hypertrophic cardiomyopathy in the United States from 1999 to 2023. Results show significant declines in mortality for both cardiomyopathies, reflecting advances in diagnostic imaging, genetic testing, and heart failure treatment. However, significant demographic and regional disparities persist. Mortality remains high among the elderly, males, and non-Hispanic Black individuals, with persistent regional differences, suggesting continued inequities in access to specialized cardiovascular care and prevention strategies. Future efforts are needed to address healthcare access and improve clinical outcomes.
TRANSLATIONAL OUTLOOK
Higher mortality rates among the elderly, males, and non-Hispanic Black individuals are largely attributed to limited access to specialized care, delayed diagnosis, and socioeconomic disadvantage. Combining population-level and clinical data can facilitate early identification of high-risk individuals, particularly in resource-poor settings. Expanding access to imaging and genetic counseling in underserved areas is key to narrowing geographic disparities. Future efforts must prioritize enhancing targeted screening programs and ensuring equitable access to advanced treatments to benefit all populations.
INTRODUCTION
Dilated cardiomyopathy (DCM) and hypertrophic cardiomyopathy (HCM) are two prevalent non-ischemic cardiomyopathies that have significant clinical and public health implications[1]. DCM, which is characterized by left ventricular dilation and systolic dysfunction, is the third most common cause of heart failure. It is associated with a high risk of arrhythmias, thromboembolic events, and sudden cardiac death[2,3]. In contrast, HCM is the most common inherited cardiomyopathy and features abnormal myocardial thickening, typically presenting as asymmetric left ventricular hypertrophy. HCM carries a significantly higher risk of cardiovascular mortality[4], including sudden cardiac death[5].
In 2010, cardiomyopathies accounted for approximately 5.9 deaths per 100,000 people globally, corresponding to approximately 403,000 deaths. These conditions remain leading indications for heart transplantation. In the United States, DCM is estimated to result in approximately 10,000 deaths and 46,000 hospitalizations annually[6]. Between 2017 and 2019, the prevalence of DCM was estimated at 118.33 cases per 100,000 people, with higher rates observed in adults aged 65 and older, males, and African Americans[7]. In comparison, HCM-related mortality remains relatively low, at less than 1%, often falling below 0.5%. Despite its relatively low annual mortality rate, attention to HCM is still warranted, particularly to identify high-risk subgroups, refine risk stratification, and develop targeted treatment plans.
Previous studies, including those based on data from the Centers for Disease Control and Prevention Wide-ranging Online Data for Epidemiologic Research (CDC WONDER) database, have examined demographic characteristics and temporal trends in DCM- and HCM-related mortality in the U.S. These studies, which reported significant declines in mortality between 1999 and 2020, found that men and non-Hispanic (NH) Black individuals experienced higher rates, with pronounced geographic disparities[8,9]. However, these studies were limited by the data available before 2020 and did not account for the potential impact of the COVID-19 pandemic. To address these gaps, the present study uses the CDC WONDER database to provide a comprehensive analysis of DCM- and HCM-related mortality trends in U.S. adults from 1999 to 2023.
METHODS
Study setting and population
Mortality data were obtained from the Wide-ranging Online Data for Epidemiologic Research (WONDER) database, maintained by the U.S. Centers for Disease Control and Prevention (CDC)[10]. CDC WONDER compiles national death certificate data from the National Center for Health Statistics, covering all 50 states and the District of Columbia. The database includes detailed demographic, geographic, and cause-of-death information, coded according to the International Classification of Diseases, Tenth Revision (ICD-10), and is commonly used in epidemiological research and public health surveillance. Since the data are publicly available and de-identified, institutional review board approval was not needed. We extracted mortality data for DCM and HCM among U.S. adults aged ≥ 25 years from 1999 to 2023. Cases were identified using ICD-10 codes I42.0 (dilated cardiomyopathy), I42.1 (obstructive hypertrophic cardiomyopathy), and I42.2 (other hypertrophic cardiomyopathy). To improve specificity and ensure accurate attribution of death to cardiomyopathy, only records in which DCM or HCM was listed as the underlying cause of death were included in the analysis.
Data abstraction
The extracted variables included year of death, sex, age group, race/ethnicity, geographic region, state, and place of death. Age was stratified into four predefined groups: 25-44, 45-64, 65-84, and ≥ 85 years. For age-stratified trend analyses, calculations were performed starting at age 25 years. Race and ethnicity were categorized according to CDC WONDER classifications as Hispanic, non-Hispanic Black, non-Hispanic White, and other races (including Asian/Pacific Islander, American Indian/Alaska Native, and multiracial individuals). Geographic regions were defined according to U.S. Census Bureau classifications: Northeast, Midwest, South, and West[11].
Statistical analysis
Age-adjusted mortality rates (AAMRs) per 100,000 population and corresponding 95% confidence intervals (CIs) were calculated using the direct standardization method, with the 2000 U.S. standard population as the reference[12]. Crude mortality rates (CMRs) were calculated as the annual number of deaths attributed to DCM or HCM divided by the corresponding annual population. Due to structural limitations of the CDC WONDER database, age-adjusted mortality rates could not be calculated separately for the age group ≥ 85 years. Therefore, only crude mortality rates were available for this group. To avoid misinterpretation, mortality estimates for individuals aged ≥ 85 years were presented descriptively and clearly labeled as crude mortality rates (CMRs). Trend analyses were restricted to age groups for which age-adjusted mortality rates could be consistently calculated.
Temporal trends in AAMRs were assessed using Joinpoint regression software (version 5.1.0.0; National Cancer Institute)[13], which fits log-linear models to identify statistically significant changes in trends over time. The optimal number of joinpoints was determined using permutation tests and the Bayesian Information Criterion (BIC) to balance model fit and parsimony. A maximum of four joinpoints was allowed to maintain interpretability. For each identified segment, the annual percentage change (APC) and corresponding 95% CI were estimated. The average annual percentage change (AAPC) for the entire study period was calculated as a weighted average of the segment-specific APCs. A slope significantly different from zero indicated a significant increasing or decreasing trend. Statistical significance was defined as a two-sided P value < 0.05. All other statistical analyses were conducted using R software (version 4.4.3).
RESULTS
Between 1999 and 2023, 101,871 adults aged 25 and older in the United States died from DCM, 64.1% of whom were male. During the same period, 24,879 adults died from HCM, 55.1% of whom were male [Tables 1 and 2].
| Measure | Deaths_1999 | AAMR_1999 | Deaths_2023 | AAMR_2023 | Start-end | APC (95%Cl) | AAPC 95%CI |
| All | 5,304 | 3.00 (2.92-3.08) | 2,798 | 1.10 (1.05-1.14) | 1999-2002 | -4.91 (-8.61 ~ -1.06) | -3.86 (-4.92 ~ -2.78) |
| 2002-2005 | 2.90 (-4.99 ~ 11.43) | ||||||
| 2005-2014 | -6.90 (-7.80 ~ -6.00) | ||||||
| 2014-2023 | -2.57 (-3.49 ~ -1.65) | ||||||
| Sex | |||||||
| Female | 1,955 | 1.92 (1.83-2.00) | 932 | 0.67 (0.63-0.72) | 1999-2001 | -10.09 (-17.50 ~ -2.01) | -4.06 (-5.33 ~ -2.78) |
| 2001-2004 | 7.00 (-2.36 ~ 17.26) | ||||||
| 2004-2013 | -7.30 (-8.24 ~ -6.35) | ||||||
| 2013-2023 | -2.98 (-3.86 ~ -2.10) | ||||||
| Male | 3,349 | 4.35 (4.20-4.50) | 1,866 | 1.57 (1.50-1.65) | 1999-2002 | -4.31 (-8.21 ~ -0.25) | -3.92 (-5.04 ~ -2.78) |
| 2002-2005 | 1.17 (-7.01-10.06) | ||||||
| 2005-2015 | -6.49 (-7.27 ~ -5.70) | ||||||
| 2015-2023 | -2.35 (-3.47 ~ -1.22) | ||||||
| Age Group | |||||||
| Age Group 25-44 | 724 | 0.85 (0.79-0.91) | 414 | 0.46 (0.42-0.50) | 1999-2006 | 0.51 (-0.87-1.91) | -2.34 (-3.08 ~ -1.59) |
| 2006-2017 | -5.45 (-6.32 ~ -4.57) | ||||||
| 2017-2023 | 0.20 (-2.07-2.53) | ||||||
| Age Group 45-64 | 1,598 | 2.65 (2.52-2.78) | 854 | 1.04 (0.97-1.11) | 1999-2005 | -2.56 (-3.72 ~ -1.38) | -3.72 (-4.66 ~ -2.78) |
| 2005-2016 | -4.83 (-5.41 ~ -4.25) | ||||||
| 2016-2021 | -0.85 (-3.57-1.95) | ||||||
| 2021-2023 | -8.06 (-16.43-1.16) | ||||||
| Age Group 65-84 | 2,326 | 7.59 (7.28-7.90) | 1,054 | 1.99 (1.87-2.11) | 1999-2002 | -8.84 (-12.87 ~ -4.61) | -5.25 (-6.54 ~ -3.94) |
| 2002-2005 | 2.41 (-7.15-12.95) | ||||||
| 2005-2012 | -9.75 (-11.41 ~ -8.07) | ||||||
| 2012-2023 | -3.30 (-4.12 ~ -2.49) | ||||||
| Age Group 85+* | 656 | 15.79 (14.58-17.00) | 476 | 7.68 (6.99-8.37) | 1999-2005 | 9.04 (3.56-14.82) | -2.16 (-4.13 ~ -0.16) |
| 2005-2014 | -8.31 (-11.30 ~ -5.21) | ||||||
| 2014-2023 | -2.89 (-6.07-0.39) | ||||||
| Race | |||||||
| Hispanic | 266 | 2.51 (2.18-2.84) | 252 | 0.78 (0.68-0.88) | 1999-2023 | -4.73 (-5.21 ~ -4.24) | -4.73 (-5.21 ~ -4.24) |
| NH Black | 1,210 | 7.00 (6.60-7.40) | 590 | 2.15 (1.97-2.32) | 1999-2005 | -2.78 (-4,81 ~ -0.71) | -4.51 (-5.43 ~ -3.58) |
| 2005-2017 | -7.65 (-8.59 ~ -6.70) | ||||||
| 2017-2023 | 0.29 (-2.62-3.30) | ||||||
| NH White | 3,686 | 2.55 (2.47-2.64) | 1,833 | 1.02 (0.97-1.07) | 1999-2005 | 0.39 (-2.24-3.09) | -3.12 (-4.23 ~ -2.00) |
| 2005-2014 | -5.97 (-7.80 ~ -4.11) | ||||||
| 2014-2023 | -2.52 (-4.42 ~ -0.59) | ||||||
| NH Other | 122 | 2.17 (1.76-2.57) | 87 | 0.52 (0.42-0.65) | 1999-2023 | -5.17 (-5.80 ~ -4.54) | -5.17 (-5.80 ~ -4.54) |
| Census Region | |||||||
| Northeast | 832 | 2.28 (2.13-2.44) | 438 | 0.93 (0.84-1.02) | 1999-2006 | -2.02 (-4.55-0.58) | -3.34 (-4.86 ~ -1.79) |
| 2006-2018 | -6.32 (-7.85 ~ -4.77) | ||||||
| 2018-2023 | 2.25 (-3.89-8.79) | ||||||
| Midwest | 1,431 | 3.41 (3.23-3.59) | 638 | 1.19 (1.10-1.29) | 1999-2007 | -1.39 (-3.16-0.41) | -3.59 (-4.80 ~ -2.37) |
| 2007-2015 | -7.16 (-9.54 ~ -4.71) | ||||||
| 2015-2023 | -2.13 (-4.58-0.38) | ||||||
| South | 1,941 | 3.08 (2.94-3.22) | 1,066 | 1.11 (1.04-1.18) | 1999-2005 | -0.27 (-2.87-2.41) | -3.93 (-5.03 ~ -2.81) |
| 2005-2013 | -8.60 (-10.78 ~ -6.36) | ||||||
| 2013-2023 | -2.24 (-3.83 ~ -0.62) | ||||||
| West | 1,100 | 3.00 (2.83-3.18) | 656 | 1.14 (1.05-1.23) | 1999-2001 | -9.09 (-18.61-1.54) | -3.61 (-5.13 ~ -2.08) |
| 2001-2004 | 3.79 (-7.25-16.13) | ||||||
| 2004-2023 | -4.15 (-4.49 ~ -3.80) |
| Measure | Deaths_1999 | AAMR_1999 | Deaths_2023 | AAMR_2023 | Start-End | APC (95%Cl) | AAPC 95%CI |
| All | 1,315 | 0.75 (0.71-0.79) | 909 | 0.35 (0.32-0.37) | 1999-2011 | -5.18 (-6.06 ~ -4.30) | -3.20 (-4.37 ~ -2.01) |
| 2011-2018 | -3.08 (-6.00 ~ -0.07) | ||||||
| 2018-2023 | 1.55 (-2.50-5.76) | ||||||
| Sex | |||||||
| Female | 546 | 0.55 (0.51-0.60) | 482 | 0.33 (0.30-0.37) | 1999-2015 | -4.72 (-5.38 ~ -4.05) | -2.30 (-3.08 ~ -1.52) |
| 2015-2023 | 2.71 (0.54-4.93) | ||||||
| Male | 769 | 0.91 (0.85-0.98) | 427 | 0.37 (0.33-0.40) | 1999-2018 | -4.73 (-5.20 ~ -4.26) | -3.60 (-4.50 ~ -2.69) |
| 2018-2023 | 0.85 (-3.51-5.40) | ||||||
| Age Group | |||||||
| Age Group 25-44 | 367 | 0.43 (0.39-0.47) | 122 | 0.14 (0.11-0.16) | 1999-2006 | -2.58 (-3.90 ~ -1.24) | -4.80 (-5.61 ~ -3.99) |
| 2006-2012 | -7.94 (-10.39 ~ -5.43) | ||||||
| 2012-2023 | -4.46 (-5.45 ~ -3.47) | ||||||
| Age Group 45-64 | 437 | 0.72 (0.66-0.79) | 229 | 0.28 (0.24-0.31) | 1999-2015 | -4.86 (-5.32 ~ -4.41) | -3.84 (-4.41 ~ -3.27) |
| 2015-2023 | -1.77 (-3.36 ~ -0.15) | ||||||
| Age Group 65-84 | 357 | 1.16 (1.04-1.29) | 383 | 0.72 (0.65-0.79) | 1999-2009 | -6.56 (-7.86 ~ -5.23) | -2.14 (-3.32 ~ -0.94) |
| 2009-2019 | -1.37 (-3.14-0.43) | ||||||
| 2019-2023 | 7.71 (2.10-13.63) | ||||||
| Age Group 85+* | 154 | 3.71 (3.12-4.29) | 175 | 2.82 (2.41-3.24) | 1999-2014 | -4.15 (-5.28 ~ -3.00) | -1.49 (-2.60 ~ -0.38) |
| 2014-2023 | 3.10 (0.55-5.71) | ||||||
| Race | |||||||
| Hispanic | 74 | 0.55 (0.42-0.71) | 78 | 0.22 (0.17-0.28) | 1999-2023 | -4.34 (-5.10 ~ -3.58) | -4.34 (-5.10 ~ -3.58) |
| NH Black | 273 | 1.48 (1.31-1.66) | 150 | 0.54 (0.45-0.63) | 1999-2014 | -7.78 (-8.56 ~ -7.00) | -4.36 (-5.22 ~ -3.48) |
| 2014-2023 | 1.64 (-0.54-3.86) | ||||||
| NH White | 930 | 0.65 (0.61-0.69) | 641 | 0.34 (0.31-0.36) | 1999-2017 | -3.91 (-4.48 ~ -3.35) | -2.70 (-3.59 ~ -1.80) |
| 2017-2023 | 1.05 (-2.42-4.65) | ||||||
| NH Other | 33 | 0.53 (0.35-0.76) | 35 | 0.22 (0.15-0.31) | - | - | - |
| Census Region | |||||||
| Northeast | 184 | 0.49 (0.42-0.56) | 176 | 0.38 (0.32-0.43) | 1999-2023 | -1.95 (-2.60 ~ -1.31) | -1.95 (-2.60 ~ -1.31) |
| Midwest | 355 | 0.86 (0.77-0.95) | 208 | 0.38 (0.33-0.44) | 1999-2010 | -6.74 (-8.12 ~ -5.34) | -3.61 (-4.51 ~ -2.70) |
| 2010-2023 | -0.88 (-2.20-0.46) | ||||||
| South | 371 | 0.58 (0.52-0.64) | 296 | 0.29 (0.25-0.32) | 1999-2015 | -5.51 (-6.47 ~ -4.53) | -3.36 (-4.51 ~ -2.20) |
| 2015-2023 | 1.07 (-2.14-4.38) | ||||||
| West | 405 | 1.08 (0.97-1.18) | 229 | 0.38 (0.33-0.43) | 1999-2019 | -5.25 (-5.85 ~ -4.64) | -4.19 (-5.54 ~ -2.82) |
| 2019-2023 | 1.28 (-6.97-10.25) |
The AAMR for DCM decreased from 3.00 (95% CI: 2.92 to 3.08) in 1999 to 1.10 (95% CI: 1.05 to 1.14) in 2023, corresponding to an AAPC of -3.86% (95% CI: -4.92 to -2.78). Mortality rates declined significantly from 1999 to 2002 (APC: -4.91%), then stabilized from 2002 to 2005. From 2005 to 2014, the decline was steeper (APC: -6.90%), followed by a slower decline from 2014 to 2023 (APC: -2.57%). For HCM, the AAMR decreased from 0.75 (95% CI: 0.71 to 0.79) in 1999 to 0.35 (95% CI: 0.32 to 0.37) in 2023, with an AAPC of -3.20% (95% CI: -4.37 to -2.01). The decline was more gradual than in DCM, with significant reductions from 1999 to 2011 (APC: -5.18%), continuing until 2018 (APC: -3.08%). A slight, non-significant increase occurred from 2018 to 2023 (APC: 1.55%) [Figure 1].
Figure 1.(A and B) show the age-stratified age-adjusted mortality rates (AAMRs, per 100,000 population) in the United States from 1999 to 2023 for DCM and HCM, respectively. The ≥ 85-year age group is not included in these charts because crude death rates, rather than age-standardized rates, were calculated for this cohort. DCM: Dilated cardiomyopathy; HCM: hypertrophic cardiomyopathy; AAMR: age-adjusted mortality rate.
Age-stratified data show that DCM- and HCM-related mortality was highest in the 65-84 age group. In the 65-84 age group, DCM exhibited an AAPC of -5.25% (95% CI: -6.54 to -3.94), while HCM showed an AAPC of -2.14% (95% CI: -3.32 to -0.94). Mortality declines were more gradual in younger age groups. For DCM in the 65-84 age group, mortality decreased sharply from 1999 to 2002 (APC: -8.84%), stabilized until 2005, then declined significantly from 2005 to 2012 (APC: -9.75%) and slowed thereafter (APC: -3.30%). For HCM in the same age group, mortality decreased significantly from 1999 to 2009 (APC: -6.56%), remained stable from 2009 to 2019 (APC: -1.37%), and showed a slight, non-significant increase from 2019 to 2023 (APC: 7.71%).
Men consistently had higher AAMRs than women for both DCM and HCM. For DCM, men experienced an AAPC of -3.92%, with mortality declining from 1999 to 2002 (APC: -4.31%), followed by a slight increase (APC: 1.17%) and then a sharp decline until 2015 (APC: -6.49%), reaching the lowest point in 2023. Women had an AAPC of -4.06%, with an initial sharp decline from 1999 to 2001 (APC: -10.09%), followed by a rise until 2004 (APC: 7.00%), and a resumed decline until 2023. For HCM, men had an AAPC of -3.60%, declining from 0.91 in 1999 to 0.37 by 2023, with a significant decrease from 1999 to 2018 (APC: -4.73%) followed by stability. In women, the AAPC was -2.30%, with a rapid decline from 1999 to 2015 (APC: -4.72%) followed by a slight increase from 2015 to 2023 (APC: 2.71%) [Figure 2].
Figure 2.(A and B) show the sex-stratified age-adjusted mortality rates (AAMRs, per 100,000 population) in the United States from 1999 to 2023 for DCM and HCM, respectively. DCM: Dilated cardiomyopathy; HCM: hypertrophic cardiomyopathy; AAMR: age-adjusted mortality rate.
The AAMRs for both DCM and HCM varied by race/ethnicity. NH Black individuals consistently had the highest AAMR, followed by NH White individuals, Hispanics, and other races. Among NH Blacks, DCM-related AAMR declined moderately from 1999 to 2005 (APC: -2.78%; 95% CI: -4.81 to -0.71). This was followed by a sharp drop from 2005 to 2017 (APC: -7.65%; 95% CI: -8.59 to -6.70), and then it stabilized through 2023 (APC: 0.29%; 95% CI: -2.62 to 3.30), resulting in an overall AAPC of -4.51% (95% CI: -5.43 to -3.58). HCM-related AAMR in NH Blacks declined from 1999 to 2014 (APC: -7.78%; 95% CI: -8.56 to -7.00) and remained stable thereafter (APC: 1.64%; 95% CI: -0.54 to 3.86), yielding an overall AAPC of -4.36% (95% CI: -5.22 to -3.48). Other racial groups saw a more gradual but consistent decline in AAMR throughout the study period, with NH Blacks consistently having the highest AAMR, followed by NH Whites, Hispanics, and other races [Figure 3].
Figure 3.(A and B) show the race/ethnicity-stratified age-adjusted mortality rates (AAMRs, per 100,000 population) in the United States from 1999 to 2023 for DCM and HCM, respectively. DCM: Dilated cardiomyopathy; HCM: hypertrophic cardiomyopathy; AAMR: age-adjusted mortality rate; NH: non-Hispanic.
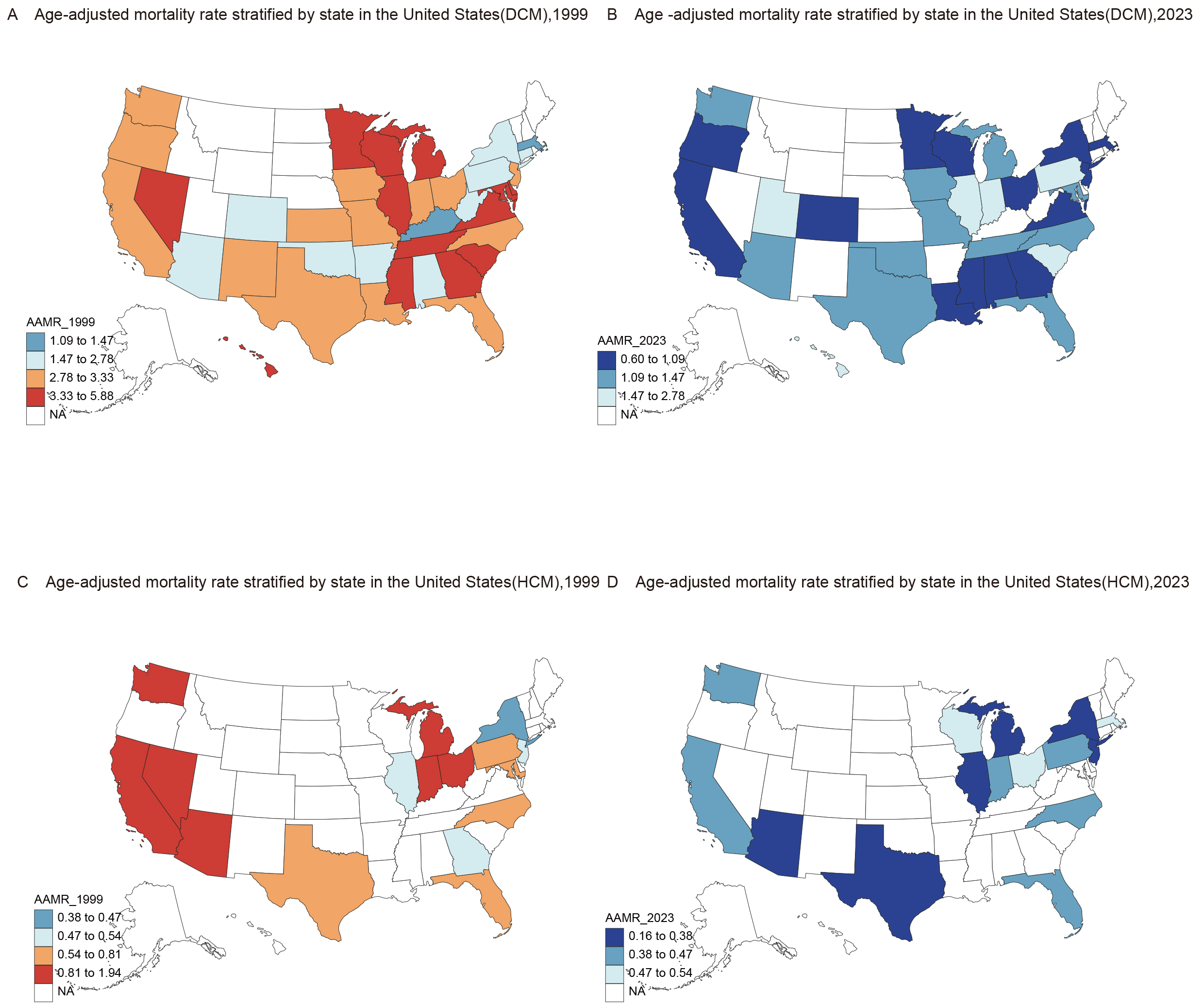
Regional differences in AAMR for DCM were evident across the United States. In the Northeast, states like New Jersey saw a significant decline in DCM mortality, with AAMR decreasing from 2.83 (95% CI: 2.39 to 3.27) in 1999 to 0.76 (95% CI: 0.57 to 1.00) in 2023. Similarly, Midwestern states, such as Wisconsin and Minnesota, demonstrated notable declines. In contrast, the Western United States had the highest AAMR for HCM, although substantial declines were observed in Nevada, where AAMR decreased from 1.93 (95%CI: 1.16 to 3.01) in 1999 to 0 in 2023. Overall, the decline in DCM mortality was more pronounced than that of HCM. By 2023, DCM AAMR had declined to 1.10 (95% CI: 1.05 to 1.14), still higher than the HCM AAMR of 0.35 (95% CI: 0.32 to 0.37). DCM-related deaths were more widely distributed across the U.S., highlighting the importance of broad surveillance and targeted public health interventions [Figures 4 and 5].
Figure 4.(A and B) show the census region–stratified age-adjusted mortality rates (AAMRs, per 100,000 population) in the United States from 1999 to 2023 for DCM and HCM, respectively. DCM: Dilated cardiomyopathy; HCM: hypertrophic cardiomyopathy; AAMR: age-adjusted mortality rate.
Figure 5.DCM-related AAMRs per 10,000, stratified by state, in adults in the United States for 1999 (A) and 2023 (B); HCM-related AAMRs per 10,000, stratified by state, in adults in the United States for 1999 (C) and 2023 (D). DCM: Dilated cardiomyopathy; HCM: hypertrophic cardiomyopathy; AAMR: age-adjusted mortality rate.
DISCUSSION
This study presents a comprehensive analysis of 25 years (1999-2023) of mortality data from the CDC WONDER database. Overall, mortality rates for both DCM and HCM declined steadily over the study period and stabilized by 2023. Mortality rates were highest among older adults, males, and non-Hispanic Black individuals. Furthermore, notable regional variations were observed. For DCM, higher AAMRs were observed in the Midwest and West, whereas in HCM, the West consistently exhibited the highest AAMRs. Together, these findings underscore the importance of demographic and regional factors in shaping mortality risk.
The observed decline in mortality may reflect cumulative advances in diagnostic and therapeutic strategies, particularly the progressive integration of evidence-based heart failure therapies in recent decades. The widespread use of echocardiography, genetic testing, and advanced imaging since the 1990s has enabled earlier detection, improved phenotyping, and more precise risk stratification, providing a foundation for individualized management[14,15]. Concurrently, the pharmacological landscape for heart failure evolved significantly from the late 1980s through the 1990s. Angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor blockers (ARBs), and β-blockers were gradually integrated into clinical practice. Subsequently, mineralocorticoid receptor antagonists (MRAs), angiotensin receptor-neprilysin inhibitors (ARNIs), and sodium-glucose cotransporter-2 inhibitors (SGLT2i) further expanded treatment options, though their uptake has been more recent and varied across populations. In DCM, these pharmacological advances, along with device-based therapies, have significantly reduced both pump-failure mortality and sudden cardiac death[16-18]. The adoption of primary prevention implantable cardioverter-defibrillators (ICDs) increased notably in the early 2000s, particularly following trials supporting their use in patients with reduced ejection fraction[16,19]. ICDs have played a crucial role in lowering arrhythmic risk in high-risk subgroups, directly impacting mortality outcomes. In contrast, for HCM, guideline-directed medical therapy has traditionally focused on symptom relief and left ventricular outflow tract obstruction management. Since most HCM patients do not have systolic dysfunction, neurohormonal therapy has not shown significant mortality benefit seen in DCM. Risk stratification for ICD implantation remains central to preventing sudden death in HCM, highlighting the unique therapeutic focus in HCM compared to DCM[15,20]. These differences in disease mechanisms and treatment strategies contribute to the distinct mortality trajectories observed in DCM and HCM.
Mortality rates for both DCM and HCM are highest among older adults. Advancing age is associated with a higher prevalence of cardiovascular comorbidities, including hypertension, atherosclerosis, and myocardial infarction. These conditions contribute to myocardial hypertrophy, reduced cardiac output, and diminished cardiac reserve, which partly explain the increased prevalence and mortality of DCM and HCM in older adults[21,22]. Additionally, older patients often have reduced tolerance to pharmacological treatments, with some medications causing adverse effects that may compromise therapeutic efficacy.
Male patients consistently exhibited higher mortality rates than female patients throughout the study period. This disparity likely reflects biological differences: estrogen provides cardioprotective effects in women, while testosterone promotes left ventricular hypertrophy in males. Premenopausal women benefit from estrogen-mediated protection, but postmenopausal declines in estrogen increase cardiovascular risk[23,24]. In contrast, higher testosterone levels in males exacerbate the risk of both DCM and HCM[25], contributing to observed sex differences in mortality[26].
Non-Hispanic Black individuals experienced the highest AAMR, driven by multiple factors, including persistent disparities in healthcare access, lower disease awareness leading to delayed treatment, and socioeconomic disadvantage[27,28]. Genetic predisposition may also contribute, as familial DCM appears more prevalent among NH Black patients[29]. The adoption of evidence-based therapies, such as sacubitril/valsartan for chronic HFrEF, which promote cardiac reverse remodeling and improved outcomes, may have contributed to the observed downward trend in mortality among NH Black patients over the study period[30].
Regional differences in mortality were pronounced. For DCM, mortality has significantly decreased in states such as Wisconsin and Minnesota in the Midwest, as well as New Jersey in the Northeast, with the lowest AAMR observed in the Northeast, likely reflecting superior healthcare infrastructure, higher socioeconomic status, and better access to specialized cardiac care. In contrast, the Western region had the highest HCM-related AAMR, highlighting regional inequities that may result from limited access to specialized care in rural and underserved areas[31]. Notably, Nevada demonstrated the most substantial reduction in AAMR among Western states, suggesting that targeted improvements in healthcare infrastructure and service delivery can mitigate regional disparities[32].
Concurrently, our study results demonstrate that the overall AAMR for DCM is significantly higher than that for HCM. This disparity in mortality burden is likely attributable to fundamental differences in the underlying etiologies and pathophysiological mechanisms of these two cardiomyopathies. DCM is characterized by progressive dilation of the cardiac chambers, particularly the left ventricle, leading to impaired systolic function. This remodeling frequently results in heart failure and malignant arrhythmias, thereby contributing directly to increased mortality. The etiology of DCM is diverse and multifactorial, encompassing genetic predispositions[33], infectious and autoimmune processes, toxic or metabolic insults (such as alcohol and chemotherapy), and environmental factors. By contrast, HCM is defined by pathological, often asymmetric, hypertrophy of the myocardium, typically involving the interventricular septum. This hypertrophy primarily compromises ventricular filling and may also impair systolic function. HCM is predominantly genetic, with the most common cause being autosomal dominant mutations in genes encoding sarcomere proteins[5,34]. Compared with HCM, the pathogenic factors underlying DCM are more diverse and complex, which may partly explain its higher overall mortality rate.
The COVID-19 pandemic substantially affected healthcare systems and cardiovascular outcomes worldwide. However, our study found no statistically significant increase in DCM and HCM mortality during the late study period. While SARS-CoV-2 infection has been associated with myocardial injury and adverse cardiac events in previous studies, the stabilization of mortality trends after 2018 does not provide evidence of a pandemic-related effect. In addition to potential myocardial injury, competing mortality, healthcare disruptions, and changes in death certification practices may have influenced cause-of-death patterns[35,36]. For instance, delays in diagnosis and treatment, along with increased mortality from COVID-19 in patients with comorbidities, may have influenced patterns of heart disease-related deaths.
This study has several important strengths in cardiomyopathy epidemiology. It extends prior national analyses by incorporating mortality data through 2023, capturing trends across the pre-pandemic period, the peak of COVID-19 disruption, and the early post-pandemic phase. Previous studies ending in 2020 could not determine whether pandemic-era fluctuations were temporary or indicated sustained shifts. Our study clarifies this, showing continued downward trends without a significant increase in HCM mortality. Additionally, this study directly compares DCM and HCM using a unified analytic framework, applying consistent case definitions, age standardization, and Joinpoint modeling. This approach enables clearer delineation of differences in mortality burden, the magnitude of decline, and demographic disparities between the two subtypes. DCM showed a more consistent and pronounced reduction in mortality compared to HCM, likely due to the greater impact of evidence-based pharmacological therapies and device interventions. In contrast, pharmacological therapy has had a more limited impact on HCM-related mortality, highlighting the continued importance of risk stratification and ICD implantation in this cohort. Furthermore, age-stratified analyses revealed variability in mortality trends across the adult lifespan, particularly among individuals aged 65-84 years, emphasizing variations in the pace and timing of mortality reduction. These findings highlight the need for age-specific strategies in managing cardiomyopathy and guide resource allocation for an aging population.
This study has several limitations. First, limitations in disease classification codes may have led to misclassification of underlying causes of death, potentially obscuring the distinction between DCM- and HCM-related mortality. Beyond classification issues, the CDC WONDER database lacks detailed information on treatment strategies, including pharmacological therapy, device implantation, and surgical interventions, limiting assessment of the impact of management approaches on mortality trends. Furthermore, the absence of key socioeconomic variables, such as education, income, insurance status, and residential environment - prevents evaluation of their contributions to disparities in outcomes.
In conclusion, while mortality rates for both DCM and HCM have declined in the U.S. population from 1999 to 2023, marked disparities persist, particularly among the elderly, men, and non-Hispanic Black individuals. These inequities are likely driven by unequal access to specialized care, broader systemic healthcare barriers, and socioeconomic disadvantages. Addressing these challenges will require expanding healthcare access, strengthening early diagnostic strategies, and implementing targeted interventions for high-risk populations to promote more equitable outcomes.
References
- Manzi L, Buongiorno F, Narciso V. Acute heart failure and non-ischemic cardiomyopathies: a comprehensive review and critical appraisal. Diagnostics (Basel).. 2025; 15:540. DOI | PubMed |
- Weintraub RG, Semsarian C, Macdonald P. Dilated cardiomyopathy. Lancet.. 2017; 390:400-14. DOI | PubMed
- Harding D, Chong MHA, Lahoti N. Dilated cardiomyopathy and chronic cardiac inflammation: Pathogenesis, diagnosis and therapy. J Intern Med.. 2023; 293:23-47. DOI
- Tuohy CV, Kaul S, Song HK, Nazer B, Heitner SB. Hypertrophic cardiomyopathy: the future of treatment. Eur J Heart Fail.. 2020; 22:228-40. DOI | PubMed
- Wang Y, Jia H, Song J. Accurate classification of non-ischemic cardiomyopathy. Curr Cardiol Rep.. 2023; 25:1299-317. DOI | PubMed |
- Mahmaljy H, Yelamanchili VS, Singhal M.PubMed
- Ababio Y, Kelly SP, Angeli FS. Prevalence and clinical burden of idiopathic dilated cardiomyopathy in the United States. Am J Med Open.. 2023; 10:100038. DOI | PubMed |
- Ashraf T, Abunada O, Kumar A. Trends in mortality and disparities in dilated cardiomyopathy across gender, race, and region in the United States (1999-2020). Ann Med Surg (Lond).. 2025; 87:627-34. DOI
- Renjihtlal SLM, Eid MM, Vyas C. Demographics and trends of hypertrophic cardiomyopathy-related mortality in the United States, 1999-2020. Curr Probl Cardiol.. 2023; 48:101681. DOI
- National Center for Health Statistics Mortality Data on CDC WONDER.
- Anderson RN, Rosenberg HM. Age standardization of death rates: Implementation of the year 2000 standard. Natl Vital Stat Rep. 1998;47:1-16, 20.DOI | PubMed
- Heymans S, Lakdawala NK, Tschöpe C, Klingel K. Dilated cardiomyopathy: causes, mechanisms, and current and future treatment approaches. Lancet.. 2023; 402:998-1011. DOI | PubMed
- Maron BJ, Desai MY, Nishimura RA. Diagnosis and evaluation of hypertrophic cardiomyopathy: JACC state-of-the-art review. J Am Coll Cardiol.. 2022; 79:372-89. DOI | PubMed
- Muhammed A, Abdelazeem M, Elewa MG, Sharief M, Ammar A. Primary prevention implantable cardioverter-defibrillator use in non-ischemic dilated cardiomyopathy based on arrhythmic risk stratification and left ventricular reverse remodeling prediction. Heart Fail Rev.. 2023; 28:229-40. DOI | PubMed |
- Limongelli G, Monda E. Implantable cardioverter defibrillator in hypertrophic cardiomyopathy: Time to avoid unnecessary procedure. Int J Cardiol.. 2022; 355:30-1. DOI | PubMed
- Desai AS, Fang JC, Maisel WH, Baughman KL. Implantable defibrillators for the prevention of mortality in patients with nonischemic cardiomyopathy: a meta-analysis of randomized controlled trials. JAMA.. 2004; 292:2874-9. DOI | PubMed
- Merlo M, Pivetta A, Pinamonti B. Long-term prognostic impact of therapeutic strategies in patients with idiopathic dilated cardiomyopathy: changing mortality over the last 30 years. Eur J Heart Fail.. 2014; 16:317-24. DOI | PubMed
- Prinz C, Farr M, Hering D, Horstkotte D, Faber L. The diagnosis and treatment of hypertrophic cardiomyopathy. Dtsch Arztebl Int.. 2011; 108:209-15. DOI | PubMed |
- Liberale L, Badimon L, Montecucco F, Lüscher TF, Libby P, Camici GG. Inflammation, aging, and cardiovascular disease: JACC review topic of the week. J Am Coll Cardiol.. 2022; 79:837-47. DOI | PubMed |
- Triposkiadis F, Xanthopoulos A, Parissis J, Butler J, Farmakis D. Pathogenesis of chronic heart failure: cardiovascular aging, risk factors, comorbidities, and disease modifiers. Heart Fail Rev.. 2022; 27:337-44. DOI | PubMed
- Hou H, Adzika GK, Wu Q. Estrogen attenuates chronic stress-induced cardiomyopathy by adaptively regulating macrophage polarizations via β(2)-adrenergic receptor modulation. Front Cell Dev Biol.. 2021; 9:737003. DOI | PubMed |
- Olivotto I, Maron MS, Adabag AS. Gender-related differences in the clinical presentation and outcome of hypertrophic cardiomyopathy. J Am Coll Cardiol.. 2005; 46:480-7. DOI
- Doleeb S, Kratz A, Salter M, Thohan V. Strong muscles, weak heart: testosterone-induced cardiomyopathy. ESC Heart Fail.. 2019; 6:1000-4. DOI | PubMed |
- Eildermann K, Goldmann S, Krause U. Differences in androgen receptor expression in human heart tissue in various types of cardiomyopathy and in aortic valve stenosis. J Cardiovasc Dev Dis.. 2023; 10:466. DOI | PubMed |
- Eberly LA, Day SM, Ashley EA. Association of race with disease expression and clinical outcomes among patients with hypertrophic cardiomyopathy. JAMA Cardiol.. 2020; 5:83-91. DOI | PubMed |
- Ntusi NAB, Sliwa K. Impact of racial and ethnic disparities on patients with dilated cardiomyopathy: JACC focus seminar 7/9. J Am Coll Cardiol.. 2021; 78:2580-8. DOI | PubMed
- Huggins GS, Kinnamon DD, Haas GJ. ; DCM Precision Medicine Study of the DCM Consortium. Prevalence and cumulative risk of familial idiopathic dilated cardiomyopathy. JAMA.. 2022; 327:454-63. DOI | PubMed |
- Ibrahim NE, Piña IL, Camacho A. ; Prospective Study of Biomarkers, Symptom Improvement and Ventricular Remodeling During Entresto Therapy for Heart Failure (PROVE-HF) Study Investigators. Racial and ethnic differences in biomarkers, health status, and cardiac remodeling in patients with heart failure with reduced ejection fraction treated with sacubitril/valsartan. Circ Heart Fail.. 2020; 13:e007829. DOI
- Singh GK, Azuine RE, Siahpush M, Williams SD. Widening geographical disparities in cardiovascular disease mortality in the United States, 1969-2011. Int J MCH AIDS. 2015;3:134-149.DOI | PubMed |
- Harrington RA, Califf RM, Balamurugan A. Call to action: rural health: a presidential advisory from the American heart association and American stroke association. Circulation.. 2020; 141:e615-44. DOI
- Eldemire R, Mestroni L, Taylor MRG. Genetics of dilated cardiomyopathy. Annu Rev Med.. 2024; 75:417-26. DOI | PubMed |
- El Hadi H, Freund A, Desch S, Thiele H, Majunke N. Hypertrophic, dilated, and arrhythmogenic cardiomyopathy: where are we? Biomedicines. 2023;11.DOI | PubMed |
- Schöley J, Aburto JM, Kashnitsky I. Life expectancy changes since COVID-19. Nat Hum Behav.. 2022; 6:1649-59. DOI | PubMed |
- Baig MFA, Chaliki K. Inpatient mortality and outcomes of COVID-19 among patients with non-ischemic cardiomyopathy: a propensity matched analysis. Heart Lung.. 2024; 68:160-5. DOI | PubMed
Published
Data Availability Statement
The authors gratefully acknowledge the Centers for Disease Control and Prevention for providing access to the database used in this study.
Issue
Section
License
Copyright (c) 2025 Qian Wang, Jinwen Chang, Yujia Jin, Chunyan Wang (Author)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



